Yale researchers develop COVID-19 drug using molecular sculpting
A collaboration between various Yale laboratories has led to the development of a compound that shows promise as a COVID-19 treatment.
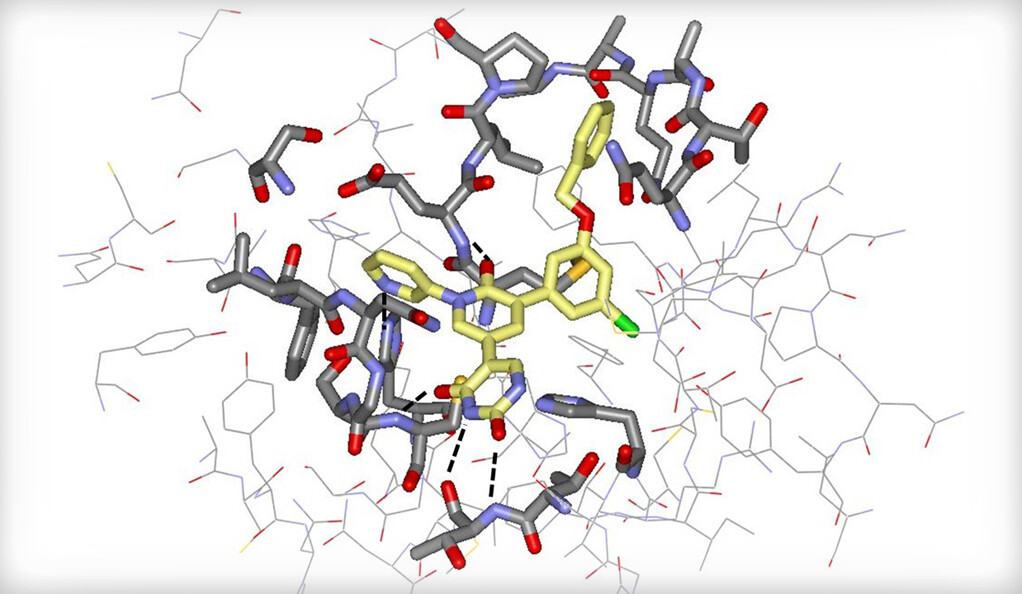
Yale News
Researchers in the Department of Chemistry and at the School of Medicine have developed a possible COVID-19 treatment using computational tools that, if implemented, could prove more resistant to new virus variants.
This new drug targets an enzyme called Mpro, a protease, or enzyme that breaks down proteins, that helps the virus replicate inside of cells. According to Sterling Professor of chemistry William Jorgensen, one of the study’s authors, the compound’s target enzyme is one of the new drug’s main advantages: Instead of the cell membrane protein spike — which is targeted by antibodies and vaccines and mutates rapidly — the enzyme targeted by this drug mutates at a much slower rate.
“It’s very important to have the type of drug that we’re developing because it’s highly likely that it will remain effective against many variants that are evolving,” Jorgensen said. “The protein that we’re targeting has a 96 percent similarity to the protease from the original coronavirus source.”
According to M.D./Ph.D. student Maya Deshmukh GRD ’25, one of the study’s authors, the study was a collaboration between the Jorgensen, Anderson, Miller, Isaacs and Lindenbach labs at Yale, spanning the Department of Chemistry and the School of Medicine. The collaboration allowed the researchers to develop the drug using computational methods and then test it in vitro.
After the drug was developed, the authors tested it in a cell culture containing cells infected with COVID-19. They found that the compound inhibited the action of Mpro and prevented the virus from continuous replication inside cells. Due to the high chances of infection from directly handling the virus, the researchers accomplished these tests using special equipment from the School of Medicine, Jorgensen said.
“The lessons learned from this experimental validation allowed us to make iterative improvements in the design of subsequent generations of compounds, arriving at promising drug candidates that are about five-thousand times more effective at inhibiting the viral protease than perampanel, the starting drug,” Deshmukh wrote in an email to the News.
According to Chun-Hui Zhang, a postdoctoral associate in the Department of Chemistry and first author on the study, the team chose the technique of molecular sculpting when developing the compound because it helps researchers quickly identify the modifications needed to improve the interactions between the drug and the protease. Through molecular sculpting, scientists can make small changes to compounds and test if it will improve its binding to a proposed target.
Deshmukh explained that the researchers saw an urgent need for a more effective drug to be coupled with the antiviral remdesivir treatment. Remdesivir is currently the only antiviral drug approved to treat COVID-19 patients but has limited efficacy when used alone.
After the article was published on Feb. 22, the Office of Cooperative Research then prepared a preliminary patent application for the drug and will now start the search for potential partners to start drug development and trials. The next step in the project, according to the authors, is testing the drug in animals to assess its safety and effectiveness before testing it in humans.
According to Jorgensen and Zhang, one of the team’s main achievements was being able to study the drug structure and develop the compound in such a short amount of time. Since the authors realized the urgency of providing a drug such as this one to the public, they collaborated in order to speed up the research and development process.
Jorgensen believes that despite the existence of protective COVID-19 vaccines, an effective treatment for the disease is still essential, since some part of the world’s population will not get vaccinated — either for personal reasons, health concerns or lack of access to the vaccine itself.
According to Deshmukh, one of the main challenges in developing treatments for the coronavirus is the appearance of new variants, against which the efficacy of some currently available vaccines is not fully proven.
“The search for new and effective direct-acting antivirals against COVID-19 must be pursued alongside global vaccination efforts to provide a therapeutic strategy for patients that do not mount an immune response to SARS-CoV-2 for a variety of reasons,” Deshmukh wrote to the News.
According to Jorgensen, the team built on years of previous research in molecular sculpting and optimization models, which the lab had previously used to develop a drug for the human immunodeficiency virus.
According to Bloomberg, the United States has administered over 300 million vaccine shots thus far.
Beatriz Horta | beatriz.horta@yale.edu








