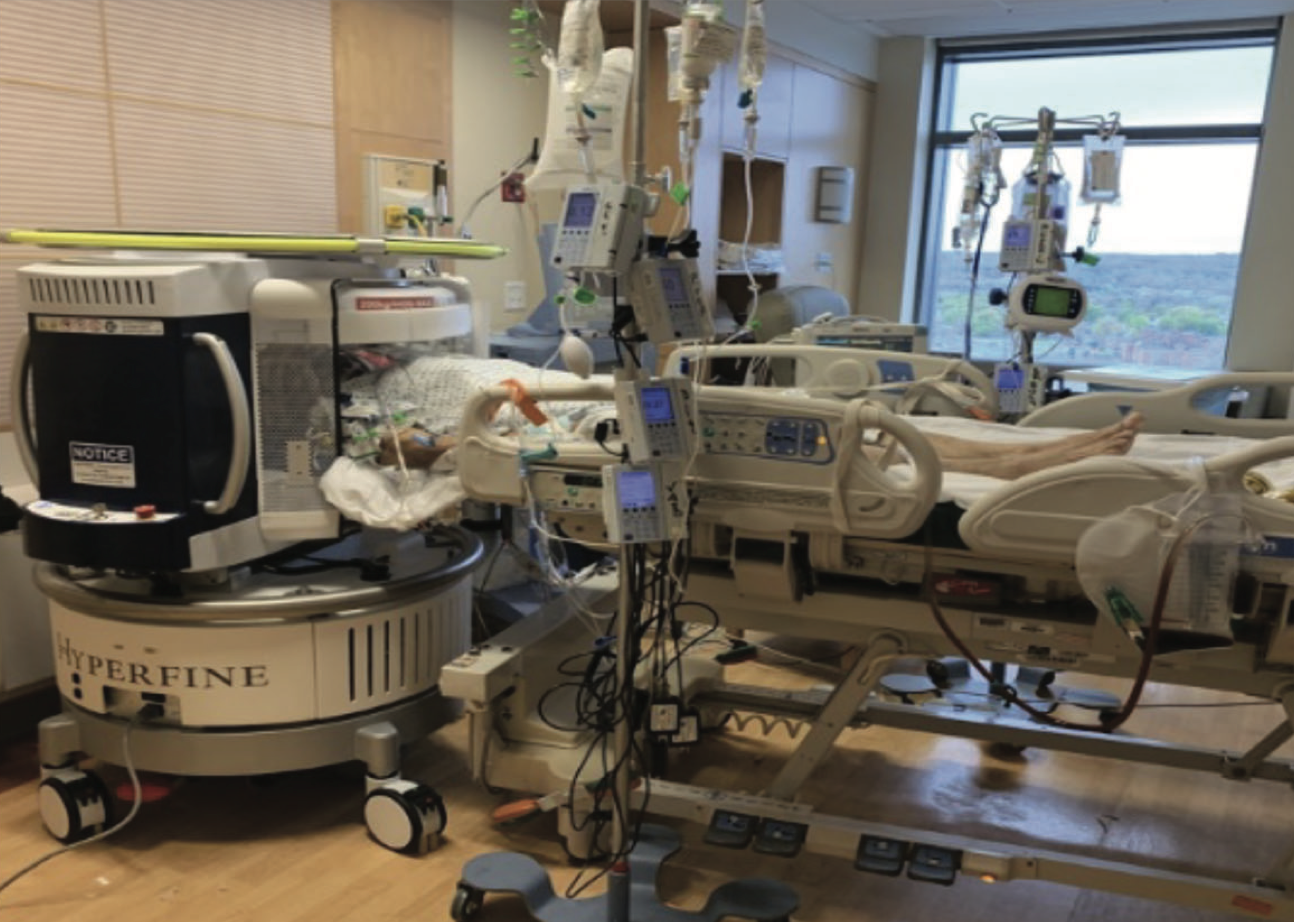
Courtesy of Hyperfine
A new portable magnetic resonance imaging device — developed by Hyperfine, a biotech company co-founded by Yale alumnus Jonathan Rothberg GRD ’91 — is being used for brain imaging at the bedside of critically ill patients in Yale New Haven Hospital’s neuroscience intensive care unit.
The device, which lends portability to the gold standard of neurological imaging, promises to revolutionize how patients in need of acute care are diagnosed for brain abnormalities. Patients in the ICU are often unconscious or hooked up to tubes and ventilators, and this complicates the process of transporting them to an imaging suite, which typically houses MRI devices due to their high magnetic field strength. But the new Hyperfine device uses a lower-strength magnetic field than the typical MRI, making it a powerful tool in dismantling barriers of transportability, access and cost. In February 2020, the device was FDA-cleared for use in clinical settings.
“The fundamental innovation here is you’re using very low field magnets, and when you have a low field magnet then the cost goes down,” said Kevin Sheth, professor of neurology and neurosurgery and YNHH chief of neurocritical care. “But also what ends up happening is the device becomes portable, it doesn’t have a strong magnet, it doesn’t need a big power supply or cooling requirement and it’s safe … so you can take it anywhere, and once you can take it anywhere, you can bring it to the bedside.”
According to Sheth, the device allows for the scanning of paralyzed and sedated patients, who are more difficult to neurologically examine. Because these patients are often in critical condition, they cannot travel outside of the ICU for imaging. The portable MRI enables physicians to look inside patients’ brains and potentially diagnose abnormalities that could otherwise go unnoticed.
Recently, a team of Yale and Harvard researchers published a paper in JAMA Neurology assessing the use of this new technology in a hospital setting. The single-center cohort study sought to evaluate whether patients in the ICU with neurological conditions could be examined using this new device. By looking at 50 patients admitted to the neuroscience and COVID-19 ICU at Yale New Haven Hospital between October 2019 and May 2020, the group was able to attain neuroimaging findings in 29 out of 30 patients who were not infected with the coronavirus. They also observed that 8 out of 20 patients who did have COVID-19 presented neurological abnormalities.
The group was led by Sheth, Massachusetts General Hospital Chief of Neurocritical Care W. Taylor Kimberly and Director of the Low-Field MRI and Hyperpolarized Media Laboratory at the Martinos Center for Biomedical Imaging Matthew Rosen. They found that the quality of images produced by the portable device was inferior to that typically yielded by regular MRI, most likely due to the lower-strength magnetic field.
But Kimberly explained that the device’s portability and lower-strength magnetic field allow it to be brought out into communities — it can run using a single standard electrical plug. The fact that the portable device enables the imaging of patients who otherwise could not be scanned made the tradeoff worthwhile, according to Kimberly.
Kimbely added that, in the same way that there are several diagnostic tools available to image different conditions, the portable MRI should add to the available methods by expanding on the possibilities and opportunities for scanning patients.
According to Kimberly, the onset of the COVID-19 pandemic exacerbated the urgency of the need for this kind of technology to diagnose neurological phenomena in comatose patients.
Kimberly told the News that when they were taking care of critically ill COVID-19 patients in their respective hospitals, he and Sheth recognized that it was difficult to assess neurological function in unconscious patients.
After some time, reports were published on potential neurological manifestations of the disease, which motivated the group to demonstrate the utility of this device in filling in this imaging void for patients in the ICU.
“This is the most valuable thing about the technology,” Kimberly said. “The fact that it’s low cost and easy to operate … opens up new directions for imaging that can enhance access to neuroimaging in a whole variety of venues, and the critically ill COVID-19 patient who is paralyzed and sedated and can’t safely move from their room is just one of many potential applications.”
In an interview with the News, Rosen — who is also co-director of the Center for Machine Learning at the Martinos Center for Biomedical Imaging, assistant professor of radiology at Massachusetts General Hospital and co-founder of Hyperfine — said that he had spent years working on ways to produce a less expensive and more transportable MRI device. This experience paved the way for his co-founding of Hyperfine with Rothberg to transform physics innovations into engineerable solutions.
Rosen dubbed the portable MRI a type of “disruptive technology,” bound to change the imaging landscape and redefine access to neuroimaging — especially in limited settings. He also described the JAMA Neurology paper as especially important in showing that it is possible to obtain adequate images from patients who might have otherwise been very difficult, if not impossible, to scan.
“Once you have ubiquitous neuroimaging, you can open up all kinds of opportunities, and … you can just get early warning and trends much quicker,” Rosen said. “That’s one of my dreams, to basically expand the places where this kind of gold standard of neuroimaging can be used, because of cost, because of ease of use, because of transportability and ubiquity.”
Sheth, Kimberly and Rosen were all enthusiastic about the device’s potential use in settings with developing health care infrastructures. Sheth described the invention as “one of the most exciting innovations that we’ve seen in years in our field.”
Rosen said that he is currently collaborating with his colleagues at the Pennsylvania State University, who study a neurological condition called postinfectious hydrocephalus, to bring the Hyperfine scanner to Uganda.
The researchers said that new projects are on the horizon to iteratively expand upon and improve clinical use of the scanner.
“In my experience in medical research and investigation, not only is it more gratifying but it always improves the research project to have a team of collaborators that each bring expertise to the table and collectively work together towards a common goal,” Kimberly said.
The Hyperfine portable MRI received FDA 510(k) clearance, which allows for the device to be used for the head imaging of patients over the age of 2.
Maria Fernanda Pacheco | maria.pacheco@yale.edu
Correction, Oct. 15: A previous version of the article stated that Rosen was collaborating with researchers at the University of Pennsylvania, instead of Pennsylvania State University.







