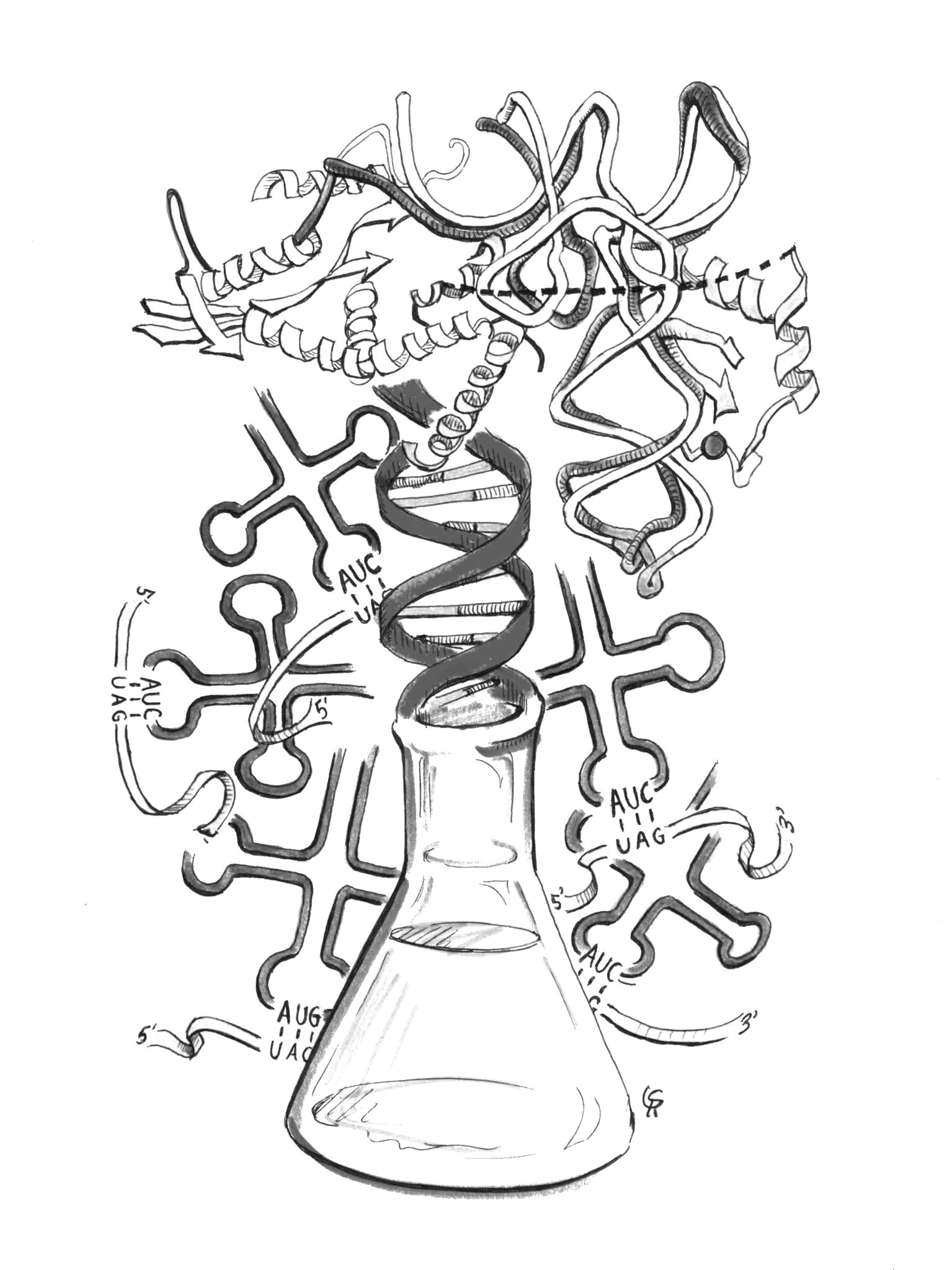
A research team at Yale has uncovered the structure of an important enzyme that could lead to the development of more effective and resistant drugs.
In a paper published Oct. 16 in Nature Chemical Biology, postdoctoral associates Tateki Suzuki and Corwin Miller presented the 3-D structure of pyrrolysyl-tRNA synthetase, a key enzyme involved in the production of synthetic proteins. A fraction of the researchers also contributed to a study published concurrently in the same journal that looked at ways to optimize the enzyme in question.
Enzymes are proteins that speed up chemical reactions; the specific enzymes the researchers studied are important in forming chains of amino acids.
“Humans are built up of proteins that typically have 20 amino acids, and so, these 20 amino acids basically accomplish the vast majority of chemistry that goes on in living organisms,” said Miller, who recently left Yale to co-found a start-up. However, he added, it is possible to modify these 20 amino acids or even incorporate additional ones. That’s where pyrrolysyl-tRNA synthetase, the researchers’ enzyme of interest, becomes relevant.
“[Pyrrolysyl-tRNA synthetase] is used to incorporate non-natural amino acids to proteins of interest,” Suzuki explained. “By incorporating non-natural amino acids, we can expand the function of proteins.”
Pyrrolysyl-tRNA synthetase captures and recruits one such non-natural amino acid, called pyrrolysine.
“What’s interesting about pyrrolysine is, since it doesn’t occur in most organisms, you can basically rewire the pyrrolysine incorporation to incorporate many other amino acids,” Miller told the News.
It is already possible to include a small number of non-natural amino acids in a protein, he said. The paper focused on techniques to integrate large numbers of these synthetic amino acids in order to design complex proteins with specific functions not seen in nature.
When scientists want to study the function of any given protein, their first step is to visualize its 3-D structure in a process called crystallization. According to Miller, understanding the structure of the enzyme used to incorporate the amino acids marks a major step forward in expanding the genetic code to include synthetic amino acids.
Describing the authors as hardworking and intelligent, senior author Dieter Soll praised the work as a unique collaboration between Suzuki, a crystallographer and Miller, a microbiologist.
“It’s marvelous to see such collaboration,” Soll said. “It really shows that a variety of expertise … is essential to drive a project forward.”
Soll added the research is not just a collaboration between disciplines but also between his lab at Yale and David Lin’s lab at Harvard. While the Yale research focused on the structure of the enzyme itself, the work at Harvard sought to design new, faster-working enzymes.
In an email to the News, Lin described the work as “a true collaboration in every sense.”
Since each study addresses a specific aspect of the enzyme, the two papers complement each other, Miller said.
The researchers interviewed gave varied responses concerning the potential applications of their work.
Soll cautioned against making any predictions about applications of the research, adding that the reach and impact of any study cannot be foreseen. He described the work’s significance factually, calling it an extremely useful tool for future research in synthetic biology.
On the other hand, Miller noted potential applications for synthetic proteins in the pharmaceutical industry.
He explained that while organisms have developed resistance to natural proteins and can break down their component amino acids, the synthetic proteins composed of many non-natural amino acids can be engineered to resist this breakdown and perform specific functions inside the body. There is potential to turn bacterial cells into drug-producing factories inside the body, he added.
However, Miller said further research must be done in the field of genetic expansion before these applications can be realized.
According to the study, previous papers have engineered pyrrolysine to be expressed in a variety of cell types, from bacterial to human.
Niki Anderson | niki.anderson@yale.edu







