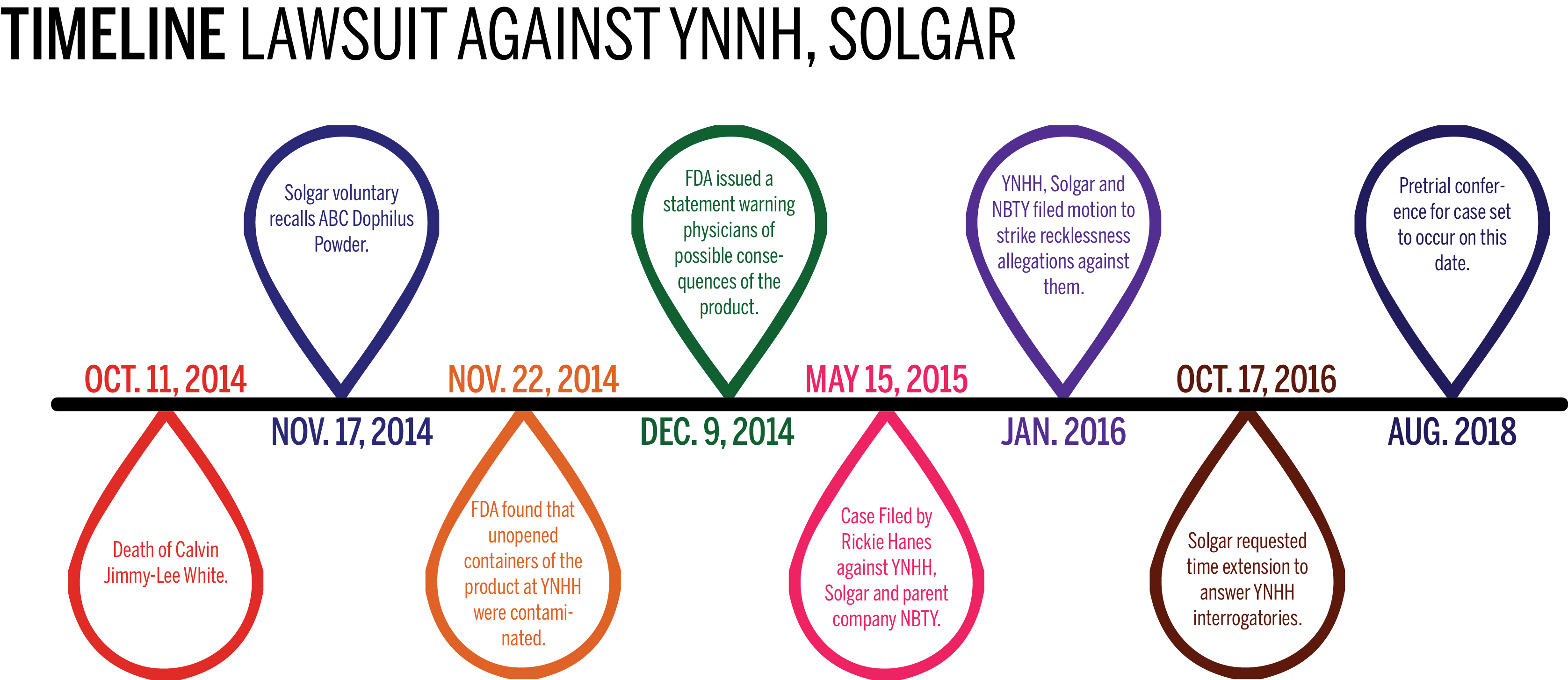
Solgar Inc., a dietary supplement manufacturing company that is Yale New Haven Hospital’s codefendant in a wrongful death lawsuit, filed a request Monday for additional time to respond to questions posed by the hospital.
The questions, issued by Yale New Haven Hospital last July, are part of the parties’ preparation for defense in a case regarding the Oct. 11, 2014, death of Calvin Lee-White eight days after his premature birth at Yale New Haven. White’s grandfather, Rickie Hanes, filed the lawsuit in May 2015 against the hospital, Solgar Inc. and Solgar’s parent company, NBTY. The lawsuit alleges that White’s death was caused by fungal contaminants in ABC Dophilus powder, a dietary supplement produced by Solgar that was given to White on several occasions after he was born.
“[Yale New Haven Hospital] knew or should have known of the risks and unknowns associated with giving an immune-compromised patient a probiotic containing live bacteria that is not tested independently prior to distribution,” the lawsuit states. “The failure of the defendant Yale New Haven Hospital to provide the decedent’s parents with relevant information regarding the risks of giving probiotics to immune compromised patients such as the decedent was a proximate cause of his death.”
ABC Dophilus is a dietary supplement intended to contain three strains of live bacteria, according to the Centers for Disease Control and Prevention’s website. The website states that these bacteria are thought to have beneficial effects in premature infants suffering from intestinal infections.
The lawsuit alleges that White’s mother, father and grandmother noticed bulges in his stomach after he was given the dietary supplement during feedings but were advised by Yale New Haven that this was a result of his prematurity.
The complaint details that the powder was contaminated at the time it left Solgar’s manufacturing plant in Leonia, New Jersey.
According to a Food and Drug Administration press release dated Nov. 17, 2014, about a month after White’s death, Solgar voluntarily recalled the powder out of an “abundance of caution” because it was found to contain Rhizopus oryzae, which is particularly harmful to premature infants.
Five days after the press release was issued, FDA scientists confirmed the presence of the fungus in unopened containers of ABC Dophilus at Yale New Haven. On Dec. 9, 2014, the FDA sent a letter alerting health care providers nationwide to the risks of using dietary supplements formulated to contain live bacteria in people with weakened immune systems.
“Dietary supplements, including those that are formulated to contain live bacteria or yeast, are generally not regulated as drugs by the FDA,” the letter stated.
The FDA’s letter also noted that the clinical trial literature did not conclusively determine the safety of treating premature infants with live bacteria.
In an email to the News, NBTY Corporate Communications Director Jodi Katz said that after numerous internal and third-party investigations, Solgar could confirm that the fungal contaminant identified in the lawsuit was not found at any point within the supply chain of ABC Dophilus powder.
“In fact, the only samples of the product that had test results reflecting the presence of the contaminant at issue, were those delivered to the FDA by the hospital pharmacy from where the supposed contaminated product was dispensed,” Katz said.
Katz added that no other serious adverse events have been reported for an ABC Dophilus product originating from the same manufacturing lot of the implicated product.
Solgar has complied with its reporting obligations to the FDA, according to Katz. Solgar’s internal investigation into the contamination of the supplement has concluded, she said.
According to a Yale New Haven Hospital statement, ABC Dophilus powder was in use at the hospital at the time that Solgar voluntarily recalled the product.
“The death of a child is tragic and our deepest sympathies go out to the family,” the statement read. “Any allegation directed to the hospital in the complaint will be defended.”
Robert Alpern, dean of the Yale School of Medicine, said that while traditionally, the FDA has not reviewed dietary supplements, the organization is now considering whether it should do so.
Alpern added that while dietary supplements fall under the purview of the FDA, reviewing them is a matter of weighing the supplements’ risks against FDA budget limitations.
“Because the FDA budget is limited — very large, but still limited — and they cover all food and drugs, they continuously need to prioritize what they review and how closely they review it,” Alpern said. “Dietary supplements have generally been viewed as safer than drugs … However, there is always the possibility that some may not be safe, and thus the FDA is considering getting more involved in this area.”
In January 2016, Solgar, NBTY and Yale New Haven Hospital filed motions to strike the recklessness allegations against them, along with the request for punitive damages asserted in the plaintiff’s revised complaint.
The defendants also moved to strike the fraudulent misrepresentation claim asserted in the complaint, as well as the plaintiff’s premature request for attorney fees.
According to a scheduling order filed this July, a pretrial conference for the case will be held in August 2018. A fixed trial date has not yet been set.







